Frankenstein’s Battery: La-BOO-ratory Manual
Frankenstein’s Battery La-BOO-ratory Manual
It’s alive, it’s allliiiiiiiiive!!! Your enthusiasm for electrochemistry that is. The La-BOO-ratory Manual, Frankenstein’s Battery, combines creativity with the principles of electrochemistry in a Halloween-themed way. Sounds like a lot, right? But this fun and simple experiment engages students to express themselves while exploring the wonderful world of science.
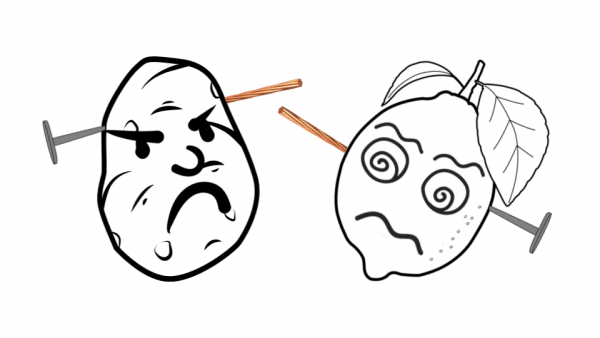
The premise of the experiment is to use pieces of produce (i.e. potatoes, lemons, etc.) to construct a monster head and then turn that monster head into an electrochemical cell. Students use markers to draw Frankenstein faces on their produce and then, with PASCO’s Wireless Voltage Sensor, detect the voltage once they insert copper wire and a galvanized nail into their monster head. The data is graphed in SPARKvue to let students visualize the voltage readings and witness science in action. Students can also continue to combine electrochemical cells and make a battery to witness how a series connection affects voltage readings.
This experiment ensures to spark students’ interests and scare them straight to a thorough understanding of electricity principles.
The way the Frankenstein Battery works is actually focused on the copper wire and galvanized nail, rather than the produce itself. The electricity comes from the electrodes made of the copper wire and the zinc-coated nail. The produce simply conducts the ions between the electrodes, so it can be called an ionic conductor. Attaching the wires completes the electrical circuit and the positive and negative ions will be conducted through the produce. The difference in electrical potential energy is what the Wireless Voltage Sensor measures and records in SPARKvue.
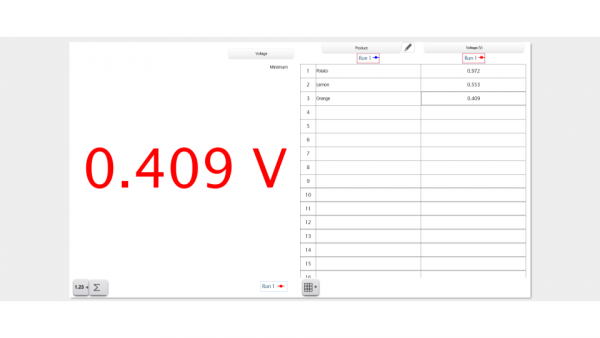
Produce that is high in potassium, sodium, and acidity make good Frankenstein Batteries due to the high amount of superconductive ions. This means the positive/negative ions that must be conducted through the produce will do so at a more efficient rate in produce that’s higher in sodium than produce that’s lower in sodium. Good Frankenstein Batteries must also have a strong internal structure for efficient conduction. This is why potatoes make such awesome Frankenstein Batteries while produce like tomatoes won’t be as great due to their messy internal structure.
Electrochemistry may seem like a monster of a topic, but engaging experiments like Frankenstein’s Battery will surely produce positive results with students.

This experiment is one of the many ways to use science to express creativity. Enabling students to create monsters and be engaged with a Halloween theme will further the combination of art into science exploration. Science is a wonder-filled world that provokes curiosity, and art is a form of unique self-expression that promotes creativity. In addition to the principles of electricity, this experiment teaches students that they don’t have to choose between creativity and curiosity.