Moving Beyond Cookie Cutter Labs
Overview
Adrian Deakin is the Science Program Area Leader and a Chemistry Teacher at Shaftesbury High School in Winnipeg. Adrian has been using PASCO equipment with his science students since 2000. Over the years he has incorporated datalogging into his science lessons and has also hosted working sessions at the local STAM conference to help familiarize other teachers with PASCO probeware.
Investigation
“I use PASCO colorimeters along with SPARK units when I am teaching my grade 11 chemistry students how to dilute solutions,” says Adrian. “We first make a kool-aid solution of known concentration and then we dilute the solution to a second known concentration and analyze it for transmittance. After converting transmittance values to absorbance we compare the absorbance readings to the proportionate change in concentration after dilution. Another grade 11 lab is the calculation of the average rates of evaporation of volatile Liquids, namely acetone, ethanol, and methanol. Students are able to monitor the temperature changes as part of the experiment. A higher level lab that I have done using colorimeters is the calculation of the percent copper in brass. Brass shell casings are dissolved in nitric acid making a solution of copper (II) ions which is tested for absorbance against a series of standard solutions. I also have students determine the concentration of ammonia in household ammonia solution via titration with hydrochloric acid. They use pH probes to monitor the pH of the solution every 1 mL upon the addition of HCl and generate a curve of pH versus volume of titrant.”
Outcome
“In the first lab, the students observe that there is a linear relationship between concentration and absorbance for coloured solutions. They also learn that transmittance values change when examined with different colours (wavelengths). Although we don’t go into further calculations, the students learn that this relationship is called Beer’s Law. In our second lab, as long as students are careful, graphs turn our very well and they can calculate the slope of the line as the substance is evaporating. It is an excellent way to introduce students to types of intermolecular forces, phase changes, and the difference between endothermic and exothermic reactions. In the advanced lab, the concentration of copper ions is used to determine the mass of copper in the sample and the percent copper is calculated by comparing the experimentally determined mass to the mass of the shell casing. These values can then be compared to typical percent composition values of brass.” These are no old-school labs!
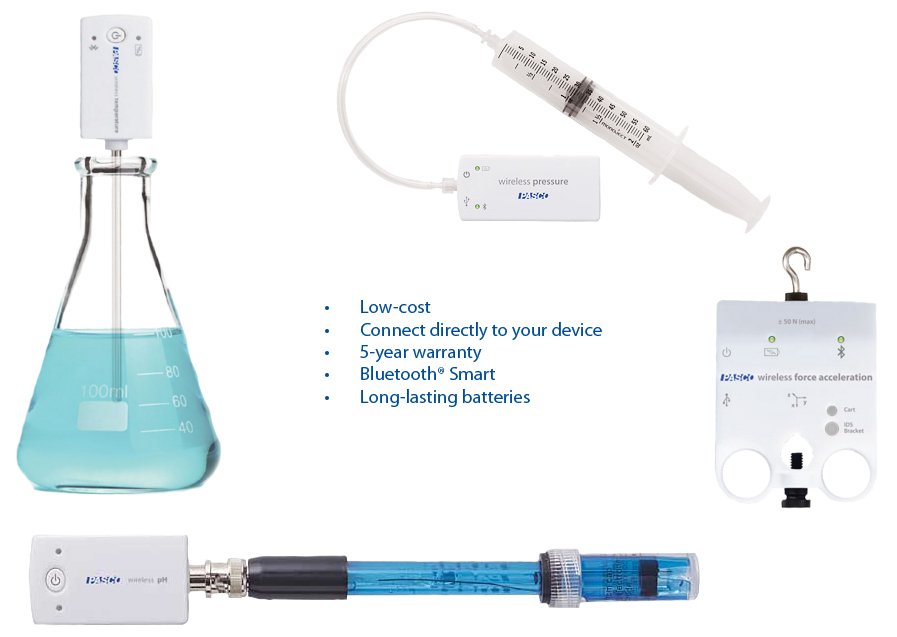
PASCO Products
- Sparkvue Software PS-2400
- Airlink PS-3200
- Colorimeter PS-2121
- pH Sensor PS-2400
- Temperature Sensor PS-3201
- Motion Sensor PS-2103A