Description
The Wireless pH Sensor is a must-have for any chemistry, biology, or environmental science course. Equally capable in the lab or field, the sensor eliminates the hassle of cables, reducing spills and improving safety. Plus, it rarely requires charging; the sensor’s coin cell battery lasts for 2-3 years in most labs and costs about one dollar to replace. It can transmit data in real time, or store data for days when continuous monitoring is required. The Wireless pH Sensor enhances countless activities, including acid-base titrations, investigations into household chemicals, analyses of chemical reactions, water quality studies, and much more.
The Wireless pH Sensor uses a BNC connector to attach the pH probe. This amplifier can be used with other probes as well, such as the ORP, Flat pH, and ISE electrodes.
— BLOG: Care & Maintenance Guide for pH Sensors —
Features
- Simplicity: just pair and go, no cables and adapters to manage
- Durable, gel-filled electrode
- Features convenient Bluetooth® wireless connectivity and long-lasting coin cell battery
- Compatible with ion-selective electrodes (ISE), oxidation-reduction probe (ORP), and Flat pH probe.
- Logging mode pH data can be recorded directly onto the sensor and later downloaded. Ideal for long-term experiments.
- Dust, dirt and sand proof and water-resistant (1 meter for 30 minutes)
Perform These Experiments
- Monitor pH during chemical reactions
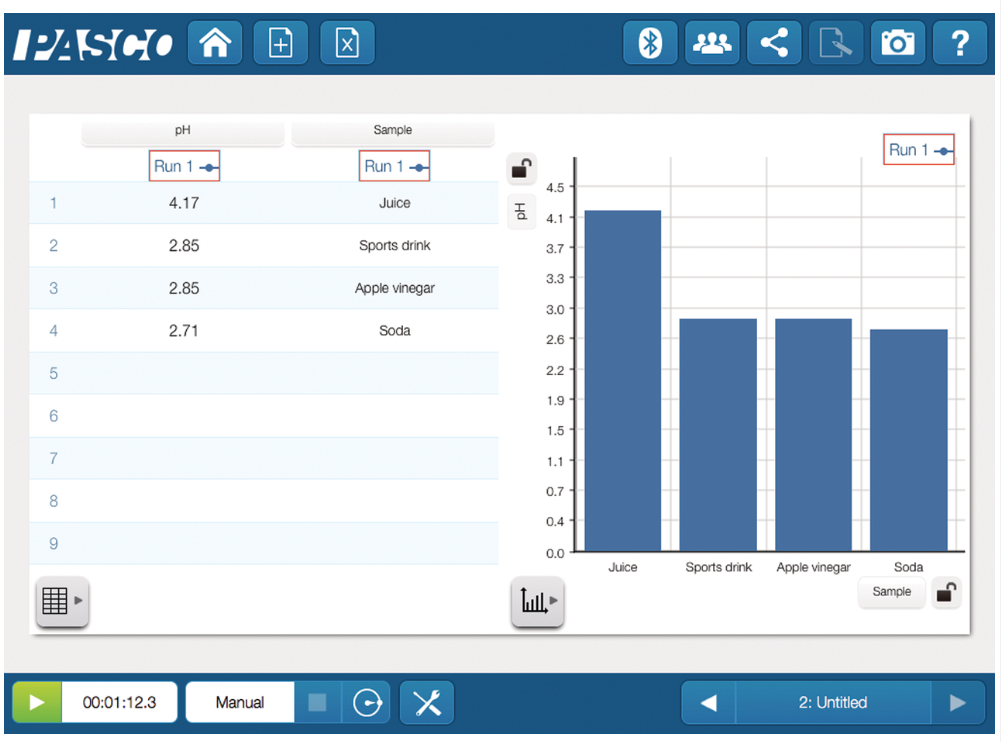
- Investigate household chemicals
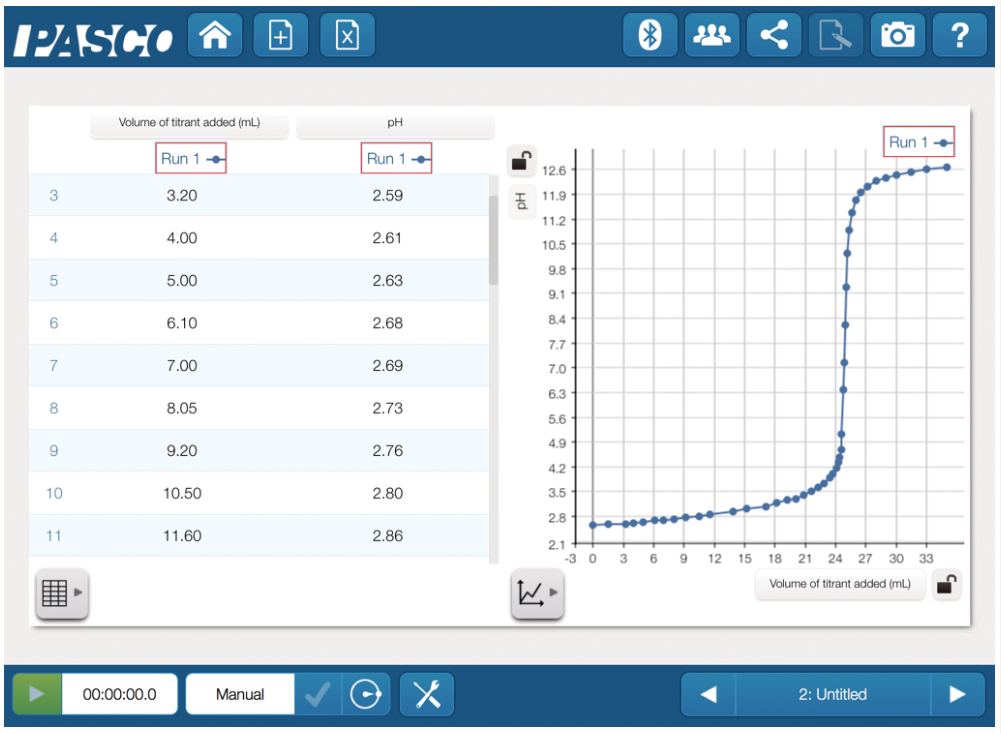
- Explore acid-base titrations
- Investigate the chemistry of buffers
- Measure pH for water quality studies
What’s Included
- 1x Coin cell battery
- 1x Direct-connect BNC pH probe
- 1x Probe storage bottle and solution
Experiment Library
Perform the following experiments and more with the Wireless pH Sensor.
Visit PASCO’s Experiment Library to view all activities for this product.
Product Specifications
| Range | 0-14 pH |
| Resolution | 0.02 pH |
| Accuracy | ±0.1 pH with calibration |
| Logging | Yes |
| Connectivity | Bluetooth 4.0 |
| Temperature Range | 5 °C to 60 °C |
Battery and Logging
| Stored Data Points Memory (Logging) 1 | >55,000 |
| Battery – Connected (Data Collection Mode) 2 | >220 hr (2-3yrs of normal classroom use)* |
| Battery – Logging (Data Logging Mode) 3 | 90 days |
| Battery Type | Coin Cell |
1 Minimum # of data points with all measurements enabled, actual results depend on enabled measurements.
2 Continuous use in a connected state until battery failure, actual results will depend on sample rate, active measurements, and battery condition.
3 Logging until battery failure, actual results will depend on sample rate, active measurements, and battery condition.
* Normal classroom use is the sensor in active use for 20min/lab for 120 lab periods/yr.
Recommended Accessories
| Storage for Wireless Temperature, pH and Conductivity Sensors | PS-3585 | |
| pH Buffer Capsule Kit | SC-2321 | |
| Electrode Support | PS-3505 | |
| Magnetic Stirrer | SE-7700 | |
| PASCO Micro Stir Bar, 5-Pack | PS-2565 |
Parts Available Separately
| Oxidation Reduction Potential Probe | PS-3515 | |
| Flat pH Probe | PS-3514 | |
| Chloride Ion Selective Electrode | PS-3519 | |
| Carbon Dioxide Ion Selective Electrode | PS-3517 | |
| Potassium Ion Selective Electrode | PS-3520 | |
| Calcium Ion Selective Electrode | PS-3518 | |
| Ammonium Ion Selective Electrode | PS-3516 | |
| Nitrate Ion Selective Electrode | PS-3521 |
Replacement Parts
| Soaker Bottle Replacement pH/ISE (5 Pack) | SE-3408 | |
| pH Electrode | PS-2573 | |
| pH Storage Solution | SC-3507 | |
| Coin Cell Battery Pack | PS-3504 |
Also Available
| Wireless Drop Counter | PS-3214 | |
| Heater Stirrer | PS-3401 | |
| USB Bluetooth Adapter | PS-3500 |
Support Documents
- Wireless pH Sensor Reference Guide
- Wireless pH sensor KCl solution
- Emergency calibration standards for pH sensors
- How do I make more pH electrode storage solution?
- How do I troubleshoot connecting a wireless sensor?
- How to install the Data Streamer app extension within Microsoft Excel
- pH Electrode Maintenance